WO1999056727A2 - Solvent/cosolvent free microemulsion and emulsion preconcentrate drug delivery systems - Google Patents
Solvent/cosolvent free microemulsion and emulsion preconcentrate drug delivery systems Download PDFInfo
- Publication number
- WO1999056727A2 WO1999056727A2 PCT/IE1999/000031 IE9900031W WO9956727A2 WO 1999056727 A2 WO1999056727 A2 WO 1999056727A2 IE 9900031 W IE9900031 W IE 9900031W WO 9956727 A2 WO9956727 A2 WO 9956727A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- agent
- composition according
- microemulsion
- oil component
- composition
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/10—Dispersions; Emulsions
- A61K9/107—Emulsions ; Emulsion preconcentrates; Micelles
- A61K9/1075—Microemulsions or submicron emulsions; Preconcentrates or solids thereof; Micelles, e.g. made of phospholipids or block copolymers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/48—Preparations in capsules, e.g. of gelatin, of chocolate
- A61K9/4841—Filling excipients; Inactive ingredients
- A61K9/4858—Organic compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/10—Dispersions; Emulsions
- A61K9/107—Emulsions ; Emulsion preconcentrates; Micelles
Definitions
- the present invention relates to pharmaceutical compositions containing a poorly water soluble therapeutic agent.
- the present invention relates to the administration, particularly oral administration to a human, of self-emulsifying microemulsion and emulsion preconcentrate formulations or microemulsions and emulsions which contain a poorly water soluble therapeutic agent, an oil component and a surfactant system.
- These preconcentrates, microemulsions and emulsions have the advantage of being substantially free of or containing only minor amounts of a hydrophilic solvent system.
- Certain oil mixtures of lipophilic drugs such as a cyclosporin, when combined with vegetable oils or other lipidic substances, surface active agents, solvents and other excipients are known to spontaneously produce dispersions of very low mean particle size, such as ⁇ 200 nm, when mixed with an agueous medium.
- These dispersions are known as microemulsions and the oily mixtures that produce the microemulsions are popularly referred to as microemulsion preconcentrates.
- the microemulsion preconcentrates are thought to produce similar dispersions of very low particle size with gastric and other physiological fluids.
- microemulsion preconcentrate formulations are known, including soft gel formulations, for enhancing the solubilization and oral bioavailability of a poorly water soluble drug compound such as cyclosporin A.
- these formulations include an active agent, an oil component, a surfactant to emulsify the formulation and a hydrophilic solvent system containing solvents/co-surfactants to solubilize the active agent.
- Typical solvents/co-surfactants include ethanol, polyethylene glycols, propylene carbonate, dimethylisosorbide, Transcutol and/or Glycofurol.
- Disadvantages of these formulations include stability or precipitation problems caused by migration of volatile hydrophilic solvents or cosolvents (e.g., ethanol can permeate a gelatin shell at normal storage temperatures), stability or precipitation problems caused by hygroscopic solvents or co-surfactants (e.g., propylene glycols, Transcutol, Glycofurol), and toxicity problems caused by addition of certain solvents or co-surfactants (e.g., dimethylisosorbide).
- volatile hydrophilic solvents or cosolvents e.g., ethanol can permeate a gelatin shell at normal storage temperatures
- hygroscopic solvents or co-surfactants e.g., propylene glycols, Transcutol, Glycofurol
- toxicity problems caused by addition of certain solvents or co-surfactants (e.g., dimethylisosorbide).
- US 5,603,951 describes a microemulsion concentrate containing cyclosporin as an active ingredient, dimethylisosorbide as a required co-surfactant, a surfactant, and an oil which can be refined fish oil, these components being present in the ratio of 1 :1-5:2-10:1-5.
- the inventors of the '951 patent added dimethylisosorbide, which is a solvent available under the Tradename ARLASOVE®, to the formulation to address the disadvantages listed above for prior solvents/co-surfactants such as ethanol, Transcutol, or Glycofurol.
- the '951 preconcentrates are formed by dissolving the cyclosporin in the dimethylisosorbide while heating at approximately 60°C followed by addition of the oil component and the surfactant.
- WO 97/33610 discloses a microemulsion preconcentrate system containing as co- surfactants propylene carbonate or a mixture of propylene carbonate with polyoxyethylene-polyoxypropylene block copolymers.
- US 5,589,455 discloses a microemulsion preconcentrate system containing the solvent polyethylene glycol.
- WO 93/20833 discloses a microemulsion preconcentrate system containing a hydrophilic phase, e.g., Transcutol, Glycofurol, 1 ,2- propylene glycol or ethanol.
- EP 760237 discloses a microemulsion preconcentrate that includes a phospholipid, preferably lecithin. Phospholipids are disadvantageous in that they are known to have stability issues.
- the oil component of a conventional microemulsion consists of fatty acid mono-, di- or thglycerides from a vegetable oil; medium chain thglycerides and/or mono- or di-glycerides; mixtures of glycerides and polyglycolized glycerides; tocol, tocopherols, and/or tocotrienols; or hydrophobic alcohols.
- WO 97/22358 discloses a microemulsions system containing tocol, tocopherols, tocotrienols and derivatives along with propylene carbonate or polyethylene glycols.
- EP 570715 discloses a microemulsion preconcentrate system containing as a lipophilic phase a mixture of polyglycolized glycerides having a HLB of less than 16.
- WO 94/25068 discloses a microemulsion preconcentrate system in which hydrophobic alcohols are used in place of the combination of hydrophilic and hydrophobic solvents; while these systems eliminate the need for a hydrophilic solvent system, the hydrophobic alcohols have the disadvantage of tasting bad.
- Cyclosporins are an example of a class of drugs that is poorly water soluble.
- Cyclosporin A is a lipophilic cyclic undecapeptide that can be isolated from the fungus Tolypoclodium inflatum Gams and which produces calcium dependent, specific and reversible inhibition of transcription of interleukin-2 and several other cytokines, most notably in T helper lymphocytes. Because of its immunosuppressive properties, it is widely used as first line therapy in the prophylaxis and treatment of transplant rejection (e.g., allo- or xeno-transplant rejection such as in patients receiving heart, lung, combined heart-lung, liver, kidney, pancreatic, skin or comeal transplants) and various autoimmune and inflammatory diseases.
- transplant rejection e.g., allo- or xeno-transplant rejection such as in patients receiving heart, lung, combined heart-lung, liver, kidney, pancreatic, skin or comeal transplants
- various autoimmune and inflammatory diseases e.g., allo-
- CyA is used in the treatment of multi-drug resistance syndrome, for example in patients undergoing chemotherapy or following organ transplantations. In patients with severe disease refractory to standard treatment; CyA is an effective therapy in acute ocular Behcet's syndrome; endogenous uveitis; psoriasis; atopic dermatitis; arthritis, particularly rheumatoid arthritis; active Crohn's disease and nephrotic syndrome.
- arthritis chronica progrediente and arthritis deformans autoimmune hematological disorders including hemolytic anemia, aplastic anemia, pure red-cell anemia and idiopathic thrombocytopenia, systemic lupus erythematosus, polychondroitis, scleroderma, Wegener granulamtosis, dermatomyositis, chronic active hepatitis, myasthenia gravis, Steven-John syndrome, idiopathic sprue, autoimmune inflammatory bowel disease, e.g., ulcerative colitis, endocrine ophthalmology, Graves disease, sarcoidosis, multiple sclerosis, primary biliary cirrhosis, juvenile diabetes, keratoconjunctivitis sicca and vernal keratoconjunctivitis, interstitial lung fibrosis, psoriatic arthritis, glomerulonephritis, juvenile dermatitis, asthma, tumors, hyper
- CyA has also been used to treat patients with moderate or severe aplastic anemia who are ineligible for bone marrow transplantation and those with primary biliary cirrhosis. CyA may be effective in patients with intractable pyoderma gangrenosum, polymyositis/dermatomyositis or severe, corticosteroid-dependent asthma. CyA is known to have a very specific effect on T-cell proliferation although the precise mechanism remains unclear. A number of non-immunosuppressive analogues of cyclosporin A have been shown to have resistance modifier activity and some are more potent than the parent compound.
- Nephrotoxicity, hepatotoxicity, hypertension, headache, hypert chosis, gingival hyperplasia, neurological and gastrointestinal effects, thrombocytopenia and microangiopathic hemolytic anemia, hyperkalemia and hyperuricemia and development of skin and lymphoproliferative malignancies are the most common adverse events in cyclosporin recipients.
- Cyclosporins are highly lipophilic, poorly water soluble and, therefore, have been supplied as an olive oil or peanut oil solution for clinical use.
- the bioavailability of cyclosporin from such oily solutions is very low and gives rise to great intersubject variation with reported systemic availability ranging from 4 to 25% (Takada, K. et al, J. Pharmacobio-Dyn., 11:80-7 (1988)).
- the bioavailability of cyclosporin has been reported to be dependent on food, bile and other interacting factors (din. Pharmacokinetics, 24:472-95 (1993)).
- CyA SAND IMMUNE® for oral administration
- SAND IMMUNE® is a solution of cyclosporin A in vegetable oil derivatives containing some other inactive excipients.
- Very high inter- and intra-patient and food dependent variability in the bioavailability of CyA has been observed from this formulation.
- the commercial microemulsion preconcentrate formulation, NEORAL® has been claimed to provide high bioavailability for CyA with low inter-and intra-patient variability.
- risks of adverse drug reactions have been indicated on switching to Neoral® (see, e.g., Drug Saf, 16:366-73 (1996); Lancet, 348:205 (1996)).
- CyA and fish oils have been administered concurrently to organ transplant patients in various clinical trials. For instance, Andreassen et al. (JAAC, 29(6): 1324-31 (1997) reported effective hypertension prophylaxis in heart transplant patients who were given cyclosporin A and 4g of fish oil. Cyclosporin A-treated and fish oil fed renal transplant recipients had improved renal function following a rejection episode (Transplantation,, 54:257 (1992)). US 5,118,493 describes the administration of CyA together with an omega-3 fatty acid oil to mediate the nephrotoxic effects of the cyclosporin.
- Omega-3 fatty acid oils possess properties that can be used for numerous therapeutic advantages, including treatment of autoimmune and inflammatory diseases such as rheumatoid arthritis, psoriasis, inflammatory bowel diseases such as Crohn's disease and ulcerative colitis; immunosuppressive treatment; hypertension prophylaxis in normal humans and in heart transplant patients; coronary heart disease; hyperlipidemia; hypertriglyceridemia; improvement of renal function and nephrotoxicity reduction.
- autoimmune and inflammatory diseases such as rheumatoid arthritis, psoriasis, inflammatory bowel diseases such as Crohn's disease and ulcerative colitis
- immunosuppressive treatment hypertension prophylaxis in normal humans and in heart transplant patients
- coronary heart disease hyperlipidemia
- hypertriglyceridemia improvement of renal function and nephrotoxicity reduction.
- omega-3 polyunsaturated fatty acids compete with omega-6 polyunsaturated fatty acids as a substrate in the arachidonic acid cascade and can therefore alter the synthesis of prostaglandin and leukotrienes, both of which are powerful mediators of inflammation and immune response.
- omega-3 fatty acid oils are described in US 5,034,415 (diabetes mellitus), US 4,843,095 (rheumatoid arthritis), JP 2253629 (anticancer), US 4,879,312 (enhancing angiogenesis), JP 1290625 (improvement of cerebral function), EP 378,824 (anti-cachexia, cholesterol and triglyceride levels reduction, platelet aggregation inhibition, colon adenocarcinomas growth inhibition), US 5,457,130 (cancer cachexia, malignant tumors, abnormal cAMP levels in adipose tissue, lipolytic activity inhibition) and US 5,436,269 (hepatitis).
- a further object is to provide self- emulsifying preconcentrates or corresponding microemulsions and emulsions having increased therapeutic agent dosing reproducibility compared to conventional formulations.
- An additional object is to provide self-emulsifying preconcentrates or corresponding microemulsions or emulsions containing a poorly water soluble therapeutic agent which provide for an enhanced amount of the therapeutic agent in a single dosage unit.
- An additional object of this invention is to provide stable self- emulsifying preconcentrates, microemulsions or emulsions containing a poorly water soluble therapeutic agent which are substantially free or contain only minor amounts of a hydrophilic solvent system. In this manner, the disadvantages of prior art systems containing hydrophilic solvent systems, such as stability problems, therapeutic agent precipitation problems and/or toxicity problems, are avoided.
- a further object of this invention is to provide a stable self-emulsifying preconcentrate and/or a microemulsion or emulsion in which the poorly water soluble therapeutic agent is substantially soluble in the oil component, thus eliminating or drastically reducing the need for substantial amounts of a hydrophilic solvent system.
- An additional object of this invention is to provide a stable self- emulsifying preconcentrate and/or a microemulsion or emulsion containing a poorly water soluble therapeutic agent and a low HLB oil component that is free or substantially free of a hydrophilic solvent system.
- a further object of this invention is to provide a stable self-emulsifying microemulsion or emulsion preconcentrate formulation and/or a microemulsion or emulsion containing a poorly water soluble therapeutic agent which is suitable for formulation into soft or hard capsules for oral administration.
- a still further object of this invention is to provide a stable self- emulsifying microemulsion or emulsion preconcentrate soft or hard capsule formulation containing an oil component and a poorly water soluble therapeutic agent having relatively high amounts of both the oil component and the poorly water soluble therapeutic agent.
- stable, self-emulsifying microemulsion or emulsion preconcentrates comprising a poorly water soluble drug can be formed from a two-component self-emulsifying system containing an oil component having an HLB ⁇ 4 and a surfactant system containing one or more surfactants, each surfactant having an HLB from about 10 to 20.
- This two-component system eliminates or drastically reduces the need for substantial amounts of a hydrophilic solvent system, allowing for formulation of preconcentrates that are substantially free of a hydrophilic solvent system or contain only minor amounts of a hydrophilic solvent system.
- the self-emulsifying microemulsion and emulsion preconcentrates according to the present invention take the form of a poorly water soluble therapeutic agent substantially solubilized in a low HLB oil component that is capable of being self-emulsified by the surfactant system when the preconcentrate is diluted with an aqueous medium.
- the present invention provides a self-emulsifying preconcentrate pharmaceutical composition suitable for administration to a mammal, particularly oral administration to a human, and capable of forming an oil-in- water microemulsion or emulsion upon dilution with an aqueous solution, comprising
- a pharmaceutically effective amount of a low HLB oil component (b) a pharmaceutically effective amount of a low HLB oil component; and (c) a surfactant system consisting essentially of at least one surfactant having an HLB of from about 10 to 20, preferably 13-19, most preferably 15-18; wherein the composition contains minor amounts or is substantially free of a hydrophilic solvent system.
- the present invention also provides microemulsions or emulsions formed by diluting a self-emulsifying preconcentrate with an aqueous solution.
- compositions according to this invention are substantially free or contain only minor amounts of a hydrophilic solvent system, the disadvantages of the prior art systems given above are avoided.
- Preferred therapeutic agents include a cyclosporin, particularly cyclosporin A, nifedipine or indomethacin and other Class 4 drugs.
- Preferred low HLB oil components include Labrafil M1944CS; Labrafil M2125CS; and fish oil or other omega-3 fatty acid oils.
- Preferred omega-3 fatty acid oils include omega-3 free fatty acids, omega-3 fatty acid thglycerides and omega- 3 fatty acid ethyl esters, such as EPA, DHA, thglycerides of EPA, thglycerides of DHA, ethyl esters of EPA, ethyl esters of DHA and mixtures thereof.
- fish oil When fish oil is employed in the compositions of the present invention, it is preferable to use fish oil containing at least 50%, preferably at least 70%, more preferably at least 80% omega-3 fatty acid oil to obtain a pharmaceutically effective amount of an omega-3 fatty acid oil in a minimal volume
- Figure 1 shows a ternary phase diagram for the placebo system described in Example 3 upon a 1 to 20 dilution of the preconcentrate with water.
- the diagram plots the relative concentration of Labrasol (0 to 100%), the concentration of the oil component Labrafil M1944CS (0 to 100%), and the concentration of Cremophor RH40 (0 to 100%) for the placebo system.
- the relative concentration of Labrasol increases from 0% at the lower right hand margin of the diagram to 100% at the lower left corner; the relative concentration of Cremophor RH40 increases from 0% at the baseline of the diagram to 100% at the apex; and the relative concentration of Labrafil M1944CS increases from 0% at the apex to 100% at the lower right hand corner of the diagram.
- the shaded area identifies those compositions having C1 , C1/C2 or C2 clarity as the microemulsion region for a 1 to 20 dilution of the preconcentrate with water;
- Figure 2 shows a ternary phase diagram for the 100 mg CyA system described in Example 3 upon a 1 to 20 dilution of the preconcentrate with water. The diagram plots the relative concentration of Labrasol (0 to 100%), the concentration of Labrafil M1944CS (0 to 100%), and the concentration of Cremophor RH40 (0 to 100%) for compositions containing 100 mg CyA.
- the relative concentration of Labrasol increases from 0% at the lower right hand margin of the diagram to 100% at the lower left corner; the relative concentration of Cremophor RH40 increases from 0% at the baseline of the diagram to 100% at the apex; and the relative concentration of Labrafil M1944CS increases from 0% at the apex to 100% at the lower right hand corner of the diagram.
- the shaded area identifies those compositions having C1 , C1/C2 or C2 clarity as the microemulsion region for a 1 to 20 dilution of the preconcentrate with water;
- Figure 3 shows a pseudo-ternary phase diagram for the placebo system described in Example 13 upon a 1 to 20 dilution of the preconcentrate with water.
- the diagram plots the relative concentration of Labrasol (0 to 100%), the concentration of the omega-3 fatty acid oil K85TG (0 to 100%), and the concentration of Cremophor RH40:Tween 80 in a 2:1 ratio (0 to 100%) for the placebo system.
- the relative concentration of Labrasol increases from 0% at the lower right hand margin of the diagram to 100% at the lower left corner; the relative concentration of Cremophor RH40:Tween 80 in a 2:1 ratio increases from 0% at the baseline of the diagram to 100% at the apex; and the relative concentration of K85TG increases from 0% at the apex to 100% at the lower right hand corner of the diagram.
- the shaded area identifies those compositions having C1 , C1/C2 or C2 clarity as the microemulsion region for a 1 to 20 dilution of the preconcentrate with water; and
- Figure 4 shows a pseudo-ternary phase diagram for the 100 mg CyA system described in Example 13 upon a 1 to 20 dilution of the preconcentrate with water.
- the diagram plots the relative concentration of Labrasol (0 to 100%), the concentration of the omega-3 fatty acid oil K85TG (0 to 100%), and the concentration of Cremophor RH40:Tween 80 in a 2:1 ratio (0 to 100%) for compositions containing 100 mg CyA.
- the relative concentration of Labrasol increases from 0% at the lower right hand margin of the diagram to 100% at the lower left corner; the relative concentration of Cremophor RH40:Tween 80 in a 2:1 ratio increases from 0% at the baseline of the diagram to 100% at the apex; and the relative concentration of K85TG increases from 0% at the apex to 100% at the lower right hand corner of the diagram.
- the shaded area identifies those compositions having C1 , C1/C2 or C2 clarity as the microemulsion region for a 1 to 20 dilution of the preconcentrate with water.
- low HLB oil component means a natural or synthetic pharmaceutically acceptable oil or mixture of oils in which the oil or mixture of oils has a hydrophile-lipophile balance (HLB) of ⁇ 4.
- Example low HLB oil components include, but are not limited to, medium and long chain (e.g., >C13) fatty acids and their mono/di/thglycerides or esters having a HLB ⁇ 4; unsaturated fatty acids and their mono/di/triglycerides or esters having a HLB ⁇ 4; and natural oils such as canoia, soybean, corn, olive, sunflower and their mono/di/triglycerides or esters having a HLB ⁇ 4; or mixtures thereof.
- medium and long chain e.g., >C13
- unsaturated fatty acids and their mono/di/triglycerides or esters having a HLB ⁇ 4 unsaturated fatty acids and their mono/di/triglycerides or esters having a HLB ⁇ 4
- natural oils
- Preferred low HLB oil components include esters, particularly the ethyl esters, of medium and long chain fatty acids.
- Specific examples of low HLB oil components include, but are not limited to, Labrafil M1944CS; Labrafil M2125CS; ethyl oleate and oleic acid; omega-6 fatty acids and their mono/di/triglycerides or esters such as ethyl esters; and fish oil and other omega-3 fatty acid oils as defined below and mixtures thereof.
- oils that are not low HLB oil components include Miglyol 812 (HLB-8-10); Gelucire 44/14 (HLB-14); Gelucire 48/09 (HLB-9); and Labrafil WO 2609 BS (HLB-6).
- omega-3 fatty acid oil means a natural or synthetic omega-3 fatty acid, and pharmaceutically acceptable esters, derivatives, precursors or salts thereof and mixtures thereof.
- omega-3 fatty acid oils include but are not limited to omega-3 polyunsaturated, long-chain fatty acids such as a eicosapenta-5, 8,11 ,14,17- enoic acid (hereinafter "EPA"), docosahexa-4,7, 10,13,16,19-enoic acid (hereinafter "DHA”), and ⁇ -linolenic acid; esters of an omega-3 fatty acid with glycerol such as mono-, di- and thglycerides; esters of the omega-3 fatty acid and a primary alcohol such as fatty acid methyl esters and fatty acid ethyl esters; precursors of an omega-3 fatty acid oil, such as EPA and DHA precursor ⁇ -linolenic acid; and derivatives such as poly
- Preferred omega-3 fatty acid oils are EPA or DHA, thglycerides thereof, ethyl esters thereof and mixtures thereof.
- the omega-3 fatty acids or their esters, derivatives, precursors, salts and mixtures thereof can be used either in their pure form or as a component of an oil such as fish oil (otherwise known as marine oil), preferably highly purified fish oil concentrates, or perilla oil or marine microalgae oil.
- Suitable fish oils are, for example, those types which are recovered in substantial quantities from cold- water fish, such as pilchard oil, menhaden oil, Peruvian fish oil, sardine oil, salmon oil, herring oil, and mackerel oil.
- the fish oil has a high omega-3 fatty acid oil content, such as 50% or higher, more preferably, 70% or higher, most preferably 80% or higher.
- suitable omega-3 fatty acid oils include the following oils available from Croda Oleochemicals (England): Incromega TG3525 (35:25 EPA:DHA ratio; thglycerides), Incromega E5015 (50:15 EPA: DHA ratio; ethyl esters) and the following oils available from Pronova Biocare (Sandefjord, Norway): EPAX6000FA, EPAX5000TG, EPAX4510TG, EPAX2050TG, K85TG, K85EE, K80EE and EPAX7010EE (further details listed in Table 1 herein).
- Preferred mixtures include mixtures of fatty acid ethyl esters and fatty acids; fatty acid ethyl esters and fatty acid thglycerides; fatty acids and fatty acid thglycerides; and fatty acid esters, fatty acid triglycerides and fatty acids such as mixtures containing K85EE and EPAX6000FA; EPAX5000TG and EPAX6000FA; K85EE and EPAX5000TG; and K85EE, EPAX6000FA and EPAX5000TG.
- the term "therapeutic agent” means a poorly water soluble drug having therapeutic use in a mammal, especially a human, or a combination of such poorly water soluble drugs wherein the drug or combination of drugs is insoluble in water or has an aqueous solubility of less than about 1 part per 1000 parts of water by weight at 20°C.
- the therapeutic agent can be selected from a variety of known types of drugs including, but not limited to, analgesics, anti-allergic agents, anti- fungals, anti-inflammatory agents, anti-arrythmic agents, antibiotics, anticoagulants, antidepressants, antidiabetic agents, anti-epilepsy agents, antihypertensive agents, anti-gout agents, anti-malarials, anti-migraine agents, antimuscarinic agents, antineoplastic agents, anti-protozoal agents, anxiolytics, thyroids, anti-thyroids, antivirals, anoretics, bisphosphonates, cardiac inotropic agents, cardiovascular agents, corticosteroids, diuretics, dopaminergic agents, gastrointestinal agents, hemostatics, histamine receptor antagonists, hypnotics, immunosuppressants, kidney protective agents, lipid regulating agents, muscle relaxants, neuroleptics, neurotropic agents, opioid agonists and antagonists, parasympathomimetics, proteas
- the therapeutic agent may comprise a combination of poorly water-soluble drugs.
- therapeutic agents include nephrotoxic drugs such as cyclosporins and amphotehcin B; indomethacin; nifedipine; cardiotoxic drugs such as amphotehcin B and FK506; drugs with immunosuppressive effects or anti-inflammatory drugs such as drugs for treating rheumatology, arthritis, psoriasis, inflammatory bowel disease, Crohn's disease or demyelinating diseases including multiple sclerosis; anti-tumor drugs such as melphalan, chlormethine, extramustinephosphate, uramustine, ifosfamide, mannomustine, trifosfamide, streptozotocin, mitobronitol, methotrexate, fluorouracil, cytarabine, tegafur, idoxide, taxol, paclitaxel, daunomycin, daunorubicin, bleomycin, amphot
- compositions according to this invention may also advantageously provide for an additive or synergistic therapeutic effect between the therapeutic agent and the omega-3 fatty acid oil or mediation of at least one negative side effect of the therapeutic agent by the omega-3 fatty acid oil.
- omega-3 fatty acid oil reduces the nephrotoxicity of cyclosporin when co-administered, allowing treatment with higher levels of cyclosporin and producing a greater clinical response at a given dose of cyclosporin.
- a pharmaceutically effective amount of a low HLB oil component means an amount effective to solubilize or substantially solubilize a therapeutically effective amount of the poorly water soluble therapeutic agent.
- substantially solubilize in reference to the solubility of the poorly water soluble therapeutic agent in the low HLB oil component means the poorly water soluble therapeutic agent is soluble in the low HLB oil component oil or has a solubility of more than 1 part per 100 parts of the low HLB oil component by weight at 20°C.
- the unit dose amount for the low HLB oil component ranges from about 5% to 70% w/w of the microemulsion or emulsion preconcentrate.
- the amount of omega-3 fatty acid oil in a unit dose of the self-emulsifying microemulsion or emulsion preconcentrate and/or microemulsion or emulsion can be adjusted so that the daily dose of the omega-3 fatty acid oil is from about 1.0 g to about 6.0 g in humans per day, preferably from about 2.0 g to about 5.0 g, most preferably about 2.5 g to about 4.0 g per day.
- the typical dosage of the omega-3 fatty acid oil ranges from about 14 to 86 mg/kg/day; the typical dosage of a fish oil contains an equivalent amount of omega-3 fatty acid oil.
- surfactant means a non-ionic or ionic surfactant having an HLB of from about 10 to 20, more preferably 13-19, most preferably 15-18.
- Suitable surfactants include but are not limited to polyoxyethylene glycolated natural or hydrogenated vegetable oils; polyoxyethylene sorbitan fatty acid esters; polyoxyethylene fatty acid esters; polyoxyethylene alkyl ethers; polyethylene glycol mono- and di- fatty acid esters; transesterification product of natural vegetable oil thglyceride with polyalkylene polyol; and fatty alcohol ethoxylates.
- Suitable surfactants include Cremophor-RH40, Cremophor-RH60, Cremophor-EL, Tween-20, Tween-40, Tween-60, Tween-65, Tween-80, Tween-85, Labrasol, Nikkol HCO-50, Nikkol HCO-40, Nikkol HCO-60, Brij 30, Gelucire 44/14, Gycerox 767, Imwitor 742, Imwitor 308, Labrafac Lipophile, Labrafac CM 10, Tagat TO, Myrj 52, Myvacet 9-45, and Vitamin E-TPGS.
- hydrophilic solvent system means a system comprising a solvent or co-solvent (other than the low HLB oil component) with respect to the poorly water soluble therapeutic agent and/or a co- surfactant having an HLB greater than about 20.
- Example hydrophilic solvent system components include ethanol, alkylene glycols such as propylene glycol, polyethylene glycol, polyoxypropylene block copolymers, Transcutol, Glycofurol, dimethylisosorbide and mixtures thereof.
- the term "minor amounts" as used in reference to a hydrophilic solvent system means an amount less than about 10% by weight of the components present in the preconcentrate, preferably less than about 5% by weight, most preferably less than the amount of therapeutic agent present in the formulation.
- the self-emulsifying microemulsion and emulsion preconcentrate and microemulsion and emulsion formulations according to the present invention may optionally include minor amounts of a hydrophilic solvent system to increase the shelf life or stability of the preconcentrates.
- Other additives, such as antioxidants or preservatives may also be present. Examples include tocopherol, tocopherols excipient, ascorbyl palmitate, butylated hydroxyanisol or other antioxidants and preservatives listed in USP XXII, Parmaceutic Ingredients.
- the self-emulsifying preconcentrates and the microemulsions and emulsions of the present invention can be adapted for oral administration.
- Preferred oral dosage forms for the preconcentrates include hard and softgel capsules.
- Preformed microemulsions and emulsions are preferred oral dosage forms for the microemulsions and emulsions.
- the formulations according to the present invention can also be administered by other routes of administration, including topical administration or parenteral administration such as i.v. or i.p. administration.
- the present invention provides for stable, self-emulsifying microemulsion or emulsion preconcentrates comprising a poorly water soluble drug formed from a two-component self-emulsifying system containing an oil component having an HLB ⁇ 4 and a surfactant system containing one or more surfactants, each surfactant having an HLB from about 10 to 20.
- This two-component system eliminates or drastically reduces the need for substantial amounts of a hydrophilic solvent system, allowing for formulation of preconcentrates that are substantially free of a hydrophilic solvent system or contain only minor amounts of a hydrophilic solvent system.
- a highly lipophilic oil component and a surfactant system containing surfactants having an HLB from about 10 to 20, preferably 13-19, more preferably 15-18 (in the absence of oils or surfactants having an HLB from about 5 to 10) provides stable preconcentrate systems.
- These particular components allow formulation of preconcentrates that are substantially free of a hydrophilic solvent system or contain only minor amounts of a hydrophilic solvent system. It is believed that the solubilizing properties of the low HLB oil component in combination with surfactants having and HLB from about 10 to 20 allow for formulation of preconcentrates that are substantially free of hydrophilic solvent system components such as surfactant or co-surfactants having an HLB > 20 or solvents.
- Enhanced bioavailability for the therapeutic agent from the preconcentrates of the present invention may be due to the absence of these hydrophilic solvent system components that could partition into the aqueous phase upon formation of the microemulsion or emulsion, causing precipitation of the therapeutic agent.
- a solution containing the poorly water soluble therapeutic agent and the oil component were prepared in appropriate proportions by adding the therapeutic agent in small increments and stirring.
- the surfactant system was prepared by mixing separately the chosen surfactants in their determined ratios.
- the oil component/therapeutic agent solution was then combined with the surfactant system solution to form the preconcentrate, such as stirring for approximately 5 minutes with or without heating to 30-40°C until homogeneous.
- formulations according to the present invention were prepared by simply combining the given amounts of the therapeutic agent, the given amounts of the oil component and the given amounts of the surfactant system with stirring until a homogeneous solution was formed.
- the therapeutic agent can be added to a mixture of the oil component and the surfactant system and stirred until a homogeneous solution was formed or the surfactant mixture can be prepared, the oil components added to it followed by addition of the drug.
- the preconcentrate was diluted, such as a 1 :1 , 1 :10, 1 :20, 1 :50 and 1 :100 v/v dilution, with an aqueous solution such as water or simulated gastric fluid to simulate conditions in the stomach.
- C1 denotes a transparent solution - particle sizing has shown that C1 systems have an average microemulsion droplet sized under 30nm;
- C2 denotes a translucent solution - droplet size is approximately 50-70nm;
- C3 denotes a slightly opaque solution - droplet size is in the region of 100nm; and
- C4 denotes a milky white solution - droplet size is much greater than 100nm.
- the self-emulsifying microemulsion systems correspond to the C1 to C2 solutions and the self- emulsifying emulsion systems correspond to the C3 to C4 solutions.
- a ternary phase diagram that maps the different clarity regions for a particular oil component surfactant system can be made to visualize the appropriate ratios needed to form a microemulsion preconcentrate or an emulsion preconcentrate formulation.
- Oral dosage forms containing the preconcentrates can be prepared.
- the therapeutic agent-containing preconcentrate can be transferred to a machine for preparing soft capsules and then encapsulated according to conventional methods for producing soft capsules.
- Samples were prepared according to Example 1 for the oil component, Labrafil M1944CS, and a surfactant system comprising Labrasol and Tween 80 with varying percentages for all three of these components.
- This system resulted in a C1 microemulsion region that contains the oil component up to approximately 20%.
- Table 1 charts the clarity values for this system (placebo) upon 1 to 20 dilution with water while Table 2 charts 1 to 20 dilution clarity values for the corresponding systems in which 25, 50, and 100 mg of the therapeutic agent cyclosporin A (CyA) per ml of solution were added.
- CyA cyclosporin A
- Samples were prepared according to Example 1 for the oil component, Labrafil M1944CS, and a surfactant system comprising Labrasol and Cremophor RH40 with varying percentages for all three of these components.
- This system resulted in a C1 or C1/C2 microemulsion region that contains the oil component up to approximately 52%, resulting in an extensive microemulsion region for these preconcentrates as shown in the ternary phase diagram in Fig. 1.
- This system offers the advantage of good clarity systems at high oil content and thus should offer good solubilization of lipophilic drugs.
- Table 3 charts the clarity values for this system (placebo) upon 1 to 20 dilution with water while Table 4 charts 1 to 20 dilution clarity values for the corresponding systems in which 25, 50, 100, 150 and 200 mg of the therapeutic agent CyA per ml of solution were added.
- C1 or C2 systems were obtained with up to 52% oil at 100 mg/ml CyA loading as shown in the ternary phase diagram of Fig. 2.
- Samples were prepared according to Example 1 for the oil component, Labrafil M1944CS, and a surfactant system comprising Tween 80 and Cremophor RH40 with varying percentages for all three of these components.
- This system resulted in a C1 or C1/C2 microemulsion region that contains the oil component up to approximately 55%, resulting in an extensive microemulsion region for these preconcentrates.
- This system offers the advantage of good clarity systems at high oil content and thus should offer good solubilization of lipophilic drugs.
- Table 5 charts the clarity values for this system (placebo) upon 1 to 20 dilution with water while Table 6 charts 1 to 20 dilution clarity values for the corresponding systems in which 25, 50, and 100 mg of the therapeutic agent CyA per ml of solution were added.
- C1 or C2 systems were obtained with up to 27% oil at 100 mg/ml CyA loading.
- Samples were prepared according to Example 1 for the oil component, Labrafil M1944CS, and a surfactant system comprising Cremophor RH40, Tween 80 and Labrasol.
- the ratio of Cremophor RH40 to Tween 80 was held constant at either 2:1 or 4:1 while the amounts of Labrasol, Labrafil and Cremophor RH40/Tween 80 were varied.
- the placebo system resulted in a C1 or C1/C2 microemulsion region that contained the oil component up to approximately 45%, resulting in an extensive microemulsion region for these preconcentrates.
- C1/C2 systems were obtained for points examined between 20% and 50% oil content.
- the placebo system results in a C1 or C1/C2 microemulsion region for up to about 45% oil content. Similar clarity systems were obtained with 50mg/ml and 100 mg/ml CyA loading. This system offers the advantage of good clarity systems at high oil content and thus should offer good solubilization of lipophilic drugs.
- the following formulation was prepared and encapsulated in softgel capsules.
- Example 6 Labrafil M1944CS / Tween 80 / Cremophor RH40 / Labrasol / Therapeutic Agents Indomethacin and Nifedipine
- Samples were prepared according to Example 1 for the oil component, Labrafil M1944CS, and a surfactant system comprising Cremophor RH40, Tween 80 and Labrasol.
- the ratio of Cremophor RH40 to Tween 80 was held constant at either 1 :1 , 1 :2, 2:1 or 4:1 while the amounts of Labrasol, Labrafil and Cremophor RH40/Tween 80 were varied.
- Table 7 and 8 describe the clarity values upon 1 to 20, 1 to 50 and 1 to 100 dilution in deionized water at 37°C for the placabo systems.
- nifedipine in microemulsion preconcentrates was examined at 25, 50 and 100 mg/g.
- Cremophor RH40 to Tween 80 ratios of 1 :2, 1:1, 2:1 and 4:1 25 mg nifedipine was fully soluble in all preconcentrates; 50 mg was solubilized in compositions with oil content up to 35%.
- Dilution of the nifedipine-loaded preconcentrate at all compositions for the 25 mg/g and at an oil content of up to 35% for the 50 mg/g nifedipine formulations produced an extensive microemulsion region with C1 or C1/C2 clarity being observed. 1 to 50 and 1 to 100 dilutions were also investigated.
- Table 10 shows the clarity values for 25 mg/g nifedipine in the 1:1 , 1:2, 2:1 and 4:1 Cremophor RH40 to Tween systems described above. Sizing of various samples in a Malvern Mastersizer S using a flow-through cell show that the visually determined C3 values correspond to a mean particle size of less than about 500 nm and C2 values correspond to a mean particle size of less than about 300 nm.
- cyclosporin A was determined in polyunsaturated omega-3 free fatty acid oil EPAX6000FA , in omega-3 fatty acid glyceride oils EPAX5000TG, EPAX4510TG, EPAX2050TG, and K85TG, in omega-3 fatty acid ethyl ester K85EE and in a mixture of free fatty acid and ethyl ester EPAX6000FA/K85EE (Pronova Biocare, Sandefjord, Norway).
- the K85TG is a mixed glyceride form obtained by transesterification of K85EE with glycerol giving the resulting blend: K85 monoglyceride: 5-15%; K85 diglyceride: 20-30%; K85 triglyceride: 50-70% and K85EE remnants: ⁇ 5%. All of these oils have an HLB ⁇ 4. Further details regarding these omega-3 fatty acid oils and the CyA solubilities are given in Table 11.
- Samples were prepared according to Example 1 for the omega-3 fatty acid oil K85EE and a surfactant system comprising Labrasol and Cremophor RH40 with varying percentages for all three of these components.
- Table 12 charts the clarity values for this system (placebo) upon 1 to 20 dilution with water while Table 13 charts 1 to 20 dilution clarity values for the corresponding systems in which 25, 50, 100 and 150 mg of CyA per ml of solution were added. From an analysis of the placebo system, it appears that the greatest amount of oil possible in a microemulsion preconcentrate formulation formulated according to this system is around 40-45% K85EE.
- Samples were prepared according to Example 1 for the omega-3 fatty acid oil K85EE and a surfactant system comprising Labrasol and Tween 80 with varying percentages for all three of these components.
- Table 14 charts the clarity values upon 1 to 20 dilution with water for this system (placebo) while Table 15 charts the 1 to 20 dilution clarity values for corresponding systems in which 25 and 50 mg of CyA per ml of solution were added.
- Samples were prepared according to Example 1 for the omega-3 fatty acid oil K85EE and a surfactant system comprising Labrasol, Tween 80 and Cremophor RH40 (holding the ratio of Cremophor RH40 to Tween 80 at 2:1) with
- Table 16 charts the 1 to 20 dilution clarity values for this system (placebo) as well as the corresponding system with 5% Ethanol included also.
- Table 17 charts 1 to 20 dilution clarity values for corresponding systems in which 100 mg of CyA per ml of solution has been added.
- Tables 16 and 17 show that inclusion of 5% ethanol compared to the same system without ethanol provides similar microemulsion region sizes for both placebo systems (20% to 50% oil) and the corresponding 100 mg/ml CyA systems.
- Samples were prepared according to Example 1 for the omega-3 fatty acid oil EPAX5000TG and a surfactant system comprising Labrasol and Cremophor RH40 with varying percentages for all three of these components.
- Table 18 charts the clarity values for this system (placebo) upon 1 to 20 dilution with water as well as 1 to 20 dilution clarity values for the corresponding systems in which 25, 50, 100 and 150 mg of CyA per ml of solution were added.
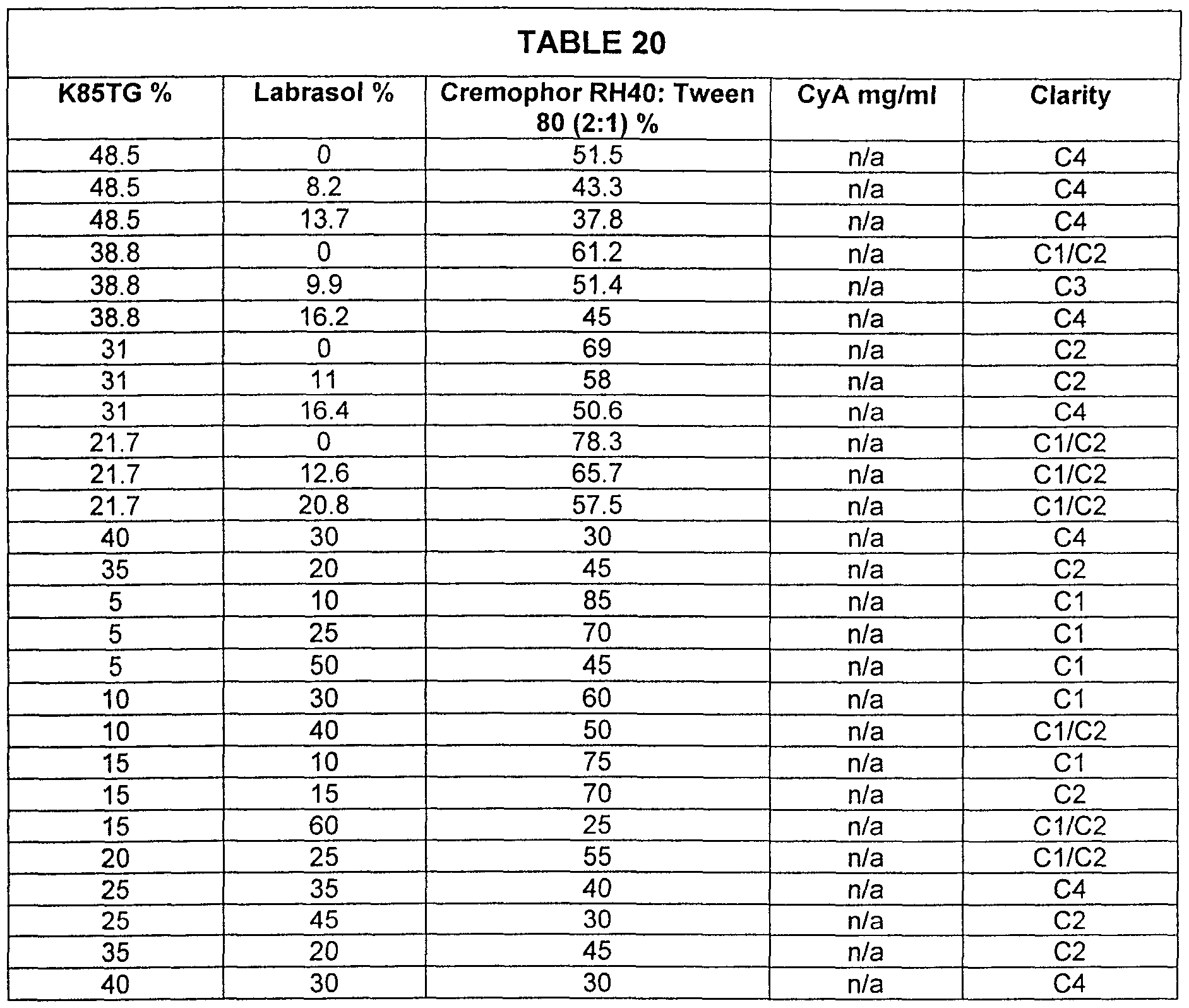
- Samples were prepared according to Example 1 for the omega-3 fatty acid oil K85TG and a surfactant system comprising Labrasol, Cremophor RH40 and Tween 80 (with Cremophor RH40 and Tween 80 held at a 2:1 ratio) with varying percentages for the oil, Labrasol and the Cremophor RH40 and Tween 80 mixture.
- Table 20 charts the clarity values for this system (placebo) upon 1 to 20 dilution with water.
- a pseudo-ternary phase diagram showing the microemulsion region (C1 , C1/C2, and C2 clarity values) for this placebo system upon 1 to 20 dilution is shown in Figure 3.
- Table 21 charts the 1 to 20 dilution clarity values for the corresponding systems in which 25, 50, 100 and 150 mg of CyA per ml of solution were added.
- the pseudo-ternary phase diagram given in Figure 4 shows the microemulsion region for the system upon 1 to 20 dilution having 100 mg/ml CyA per ml of solution.
- Samples were prepared according to Example 1 for the omega-3 fatty acid oil K85TG and a surfactant system comprising Labrasol and Cremophor RH40 with varying percentages for all three of these components.
- Table 22 charts the clarity values for this system (placebo) upon 1 to 20 dilution with water as well as 1 to 20 dilution clarity values for the corresponding systems in which 25, 50, 100 and 150 mg of CyA per ml of solution were added. From an analysis of the placebo system, it appears that the greatest amount of oil possible in a microemulsion preconcentrate formulation formulated according to this system is around 27% K85TG.
- microemulsion region on a ternary phase diagram obtained by plotting the data for the K85EE/Cremophor/Labrasol system provided in this example is similar to that for the corresponding EPAX5000TG system (Example 11) and the EPAX6000FA system (Example 12) over a range of 0 to 150 mg/ml CyA.
- the corresponding K85EE system (Example 8) appears to form a larger microemulsion region than the K85TG system.
- Samples were prepared according to Example 1 for the systems containing a mixture of K85EE and EPAX6000FA and a surfactant system comprising Labrasol, Tween 80 and Cremophor RH40 with varying percentages as described in Table 23.
- Table 23 charts the clarity values for these systems (placebo) upon 1 to 20 dilution with water as well as 1 to 20 dilution clarity values for the corresponding systems in which 50 or 100 mg of CyA per ml of solution were added. There was a slight improvement in the clarity of the resulting microemulsions upon the addition of CyA.
- the following formulations according to the present invention were prepared as follows.
- the given amounts of cyclosporin, the given amounts of the oil containing omega-3 fatty acid oil, and the given amounts of the surfactant system were stirred until a homogeneous solution was formed.
- the resulting cyclospo n-containing composition was transferred to a machine for preparing soft capsules and then encapsulated according to conventional methods for producing soft capsules.
- Preconcentrate formulations suitable for formulation with a therapeutically effective amount of a poorly water soluble theraputic agent according to this invention and containing ethyl oleate were prepared as detailed in Table 24 (w/w %).
- the respective amounts of Cremophor RH40 and Tween 80 (2:1) were weighed out initially and allowed to mix at approximately 37°C until homogenous. Following this, Labrasol was added as required and all three surfactants were allowed combine until they had formed a clear homogenous mix. The appropriate oil component was added to the mixture of surfactants and allowed to mix. Finally, ethanol was included if present.
- Microemulsions were formed by diluting 1 :20 with deionised water and microemulsion formation was assessed. The average particle size was measured by photon correlation spectroscopy (C3 and C4 formulations diluted up to 1:400 as necessary) as shown in Table 24.
- Labrasol composition was found to influence droplet size, which increased to 74.5nm and 113.3nm for a Labrasol concentration of 5% and 10% respectively.
- a hazy preconcentrate with a C4 microemulsion of droplet size 135.8nm was produced.
- preconcentrate formulations suitable for formulation with a therapeutically effective amount of a poorly water soluble theraputic agent according to this invention and containing oleic acid were prepared and assessed as detailed in Table 25 (w/w %).
- the preconcentrates produced were clear, one-phased systems on formulation, and showed no sign of phase separation or streaking after being left to stand for 24hrs.
- Dilution (1 in 20) of the 15% and 30% oleic acid preconcentrates gave a clarity of C4 and an average particle size of 194.0nm and 211.3nm respectively.
- Concentrations of 37% and 50% oleic acid were used to make up preconcentrates, in conjunction with 5% Labrasol, 5% ethanol and Cremopho ⁇ Tween (2:1) as required. Both formulations gave one phase, clear, pale yellow preconcentrates.
Abstract
Description
Claims
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CA002331640A CA2331640A1 (en) | 1998-05-07 | 1999-05-07 | Solvent/cosolvent free microemulsion and emulsion preconcentrate drug delivery systems |
| EP99921098A EP1075252A2 (en) | 1998-05-07 | 1999-05-07 | Solvent/cosolvent free microemulsion and emulsion preconcentrate drug delivery systems |
| JP2000546754A JP2002513750A (en) | 1998-05-07 | 1999-05-07 | Solvent / co-solvent free microemulsion and emulsion pre-concentrate drug delivery system |
| AU38439/99A AU3843999A (en) | 1998-05-07 | 1999-05-07 | Solvent/cosolvent free microemulsion and emulsion preconcentrate drug delivery systems |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US8451898P | 1998-05-07 | 1998-05-07 | |
| US60/084,518 | 1998-05-07 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO1999056727A2 true WO1999056727A2 (en) | 1999-11-11 |
| WO1999056727A3 WO1999056727A3 (en) | 2000-03-02 |
Family
ID=22185451
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IE1999/000031 WO1999056727A2 (en) | 1998-05-07 | 1999-05-07 | Solvent/cosolvent free microemulsion and emulsion preconcentrate drug delivery systems |
Country Status (5)
| Country | Link |
|---|---|
| EP (1) | EP1075252A2 (en) |
| JP (1) | JP2002513750A (en) |
| AU (1) | AU3843999A (en) |
| CA (1) | CA2331640A1 (en) |
| WO (1) | WO1999056727A2 (en) |
Cited By (51)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2001028520A1 (en) * | 1999-10-20 | 2001-04-26 | Vesifact Ag | Microemulsion preconcentrates, microemulsion and use of such a composition |
| WO2001066087A1 (en) * | 2000-03-08 | 2001-09-13 | Astrazeneca Ab | New self emulsifying drug delivery system |
| WO2001066088A1 (en) * | 2000-03-08 | 2001-09-13 | Astrazeneca Ab | New self emulsifying drug delivery system |
| EP1158959A1 (en) | 1999-02-26 | 2001-12-05 | Lipocine, Inc. | Compositions and methods for improved delivery of hydrophobic therapeutic agents |
| EP1170003A1 (en) * | 2000-07-07 | 2002-01-09 | Hovid SDN BHD | Self-emulsifying drug delivery system for fat-soluble drugs |
| FR2818905A1 (en) * | 2000-12-28 | 2002-07-05 | Cll Pharma | MICELLAR COLLOIDAL PHARMACEUTICAL COMPOSITIONS COMPRISING A LIPOPHILIC ACTIVE INGREDIENT |
| EP1249231A1 (en) * | 2001-04-12 | 2002-10-16 | Vesifact Ag | Pharmaceutical formulations comprising anti-inflammatory compounds and use thereof |
| WO2003055466A1 (en) * | 2001-12-27 | 2003-07-10 | Cj Corporation | Microemulsion preconcentrate |
| AU771439B2 (en) * | 2000-06-08 | 2004-03-25 | Berhad, Hovid | A novel drug delivery system : formulation for fat-soluble drugs |
| JP2004517052A (en) * | 2000-10-11 | 2004-06-10 | セフアロン・インコーポレーテツド | Modafinil compound-containing composition |
| WO2004082625A2 (en) * | 2003-03-18 | 2004-09-30 | Advanced Medical Optics, Inc. | Self-emulsifying compositions, methods of use and preparation |
| EP1466587A1 (en) * | 2003-04-11 | 2004-10-13 | L'oreal | Composition in the form of an oil-in-water emulsion and its cosmetic uses |
| JP2005504788A (en) * | 2001-09-07 | 2005-02-17 | アストラゼネカ・アクチエボラーグ | Novel self-emulsifying drug delivery system |
| WO2005102277A1 (en) * | 2004-04-19 | 2005-11-03 | Hewlett-Packard Development Company, L.P. | A composition of a jetable microemulsion, a method of manufacturing thereof as well as a system of dispensing |
| JP2006241167A (en) * | 2001-02-11 | 2006-09-14 | Aquanova German Solubilisate Technologies (Agt) Gmbh | Method for producing active ingredient concentrate and active ingredient concentrate |
| EP1762557A2 (en) | 2000-02-16 | 2007-03-14 | The Brigham And Women's Hospital, Inc. | Aspirin-triggered lipid mediators |
| WO2008002121A2 (en) * | 2006-06-28 | 2008-01-03 | Hovid, Berhad | A self-emulsifying composition for poorly bioavailable drugs |
| FR2925855A1 (en) * | 2007-12-26 | 2009-07-03 | Virbac Sa Sa | FLORFENICOL SELF-CONTAINING LIQUID COMPOSITION FOR INCORPORATING BEVERAGE WATER IN ANIMAL BREEDING ANIMALS. |
| US7582785B2 (en) | 2002-04-01 | 2009-09-01 | University Of Southern California | Trihydroxy polyunsaturated eicosanoid derivatives |
| US7585856B2 (en) | 2002-08-12 | 2009-09-08 | The Brigham And Women's Hospital, Inc. | Resolvins: Biotemplates for novel therapeutic interventions |
| US7741368B2 (en) | 2001-12-18 | 2010-06-22 | The Brigham And Women's Hospital, Inc. | Approach to antimicrobial host defense with molecular shields with EPA and DHA analogs |
| US7759395B2 (en) | 2002-08-12 | 2010-07-20 | The Brigham And Women's Hospital, Inc. | Use of docosatrienes, resolvins and their stable analogs in the treatment of airway diseases and asthma |
| WO2010103402A1 (en) * | 2009-03-09 | 2010-09-16 | Pronova Biopharma Norge As | Compositions comprising a fatty acid oil mixture comprising epa and dha in free acid form and a surfactant, and methods and uses thereof |
| US7842791B2 (en) | 2002-12-19 | 2010-11-30 | Nancy Jean Britten | Dispersible pharmaceutical compositions |
| US7879345B2 (en) | 2003-04-11 | 2011-02-01 | L'oreal | Composition containing an amphiphilic polymer, uses thereof |
| WO2012032414A2 (en) * | 2010-09-08 | 2012-03-15 | Pronova Biopharma Norge As | Compositions comprising a fatty acid oil mixture, a surfactant, and a statin |
| EP2433630A1 (en) * | 2009-05-22 | 2012-03-28 | Mochida Pharmaceutical Co., Ltd. | Self-emulsifying composition of 3 fatty acid |
| WO2012032417A3 (en) * | 2010-09-08 | 2012-05-03 | Pronova Biopharma Norge As | Compositions comprising a fatty acid oil mixture, a free fatty acid, and a statin |
| WO2012032415A3 (en) * | 2010-09-08 | 2012-05-03 | Pronova Biopharma Norge As | Compositions comprising a fatty acid oil mixture comprising epa and dha in free acid form, a surfactant, and a statin |
| EP2490678A1 (en) * | 2009-10-23 | 2012-08-29 | Pronova BioPharma Norge AS | Coated capsules and tablets of a fatty acid oil mixture |
| US8299116B2 (en) | 2002-07-30 | 2012-10-30 | Wyeth Llc | CCI-779 concentrate formulations |
| RU2480208C2 (en) * | 2006-10-04 | 2013-04-27 | М Унд П Патент Акциенгезелльшафт | Delivery system with controlled release for nasal application of neurotransmitters |
| US8518961B2 (en) | 2004-11-19 | 2013-08-27 | Sigma-Tau Industrie Farmaceutiche Riunite S.P.A. | Pharmaceutical compositions comprising a camptothecin derivate |
| EP2298284A3 (en) * | 2001-02-14 | 2013-12-18 | GW Pharma Limited | Mucoadhesive pharmaceutical formulations |
| US8728518B2 (en) | 2004-08-27 | 2014-05-20 | Cspc Zhongqi Pharmaceutical Technology (Shijiazhuang) Co., Ltd | Butylphthalide self-emulsifying drug delivery system, its preparation and method and application |
| US20140287004A1 (en) * | 2005-11-14 | 2014-09-25 | The Trustees Of Columbia University In The City Of New York | Use of an Omega-3 Lipid-Based Emulsion Following Ischemic Injury to Provide Protection and Recovery in Human Organs |
| US8932610B2 (en) | 2010-03-01 | 2015-01-13 | Laboratorios Salvat, S.A. | Aqueous clear solutions of fluocinolone acetonide for treatment of otic inflammation |
| US8940786B2 (en) | 2012-10-01 | 2015-01-27 | Teikoku Pharma Usa, Inc. | Non-aqueous taxane nanodispersion formulations and methods of using the same |
| US20150272921A1 (en) * | 2005-11-14 | 2015-10-01 | The Trustees Of Columbia University In The City Of New York | Use of an Omega-3 Lipid-Based Emulsion Following Ischemic Injury to Provide Protection and Recovery in Human Organs |
| WO2015198350A1 (en) * | 2014-06-25 | 2015-12-30 | Synergia Bio Sciences Private Limited | A pharmaceutical oil-in-water nano-emulsion |
| US9566237B2 (en) | 2002-11-01 | 2017-02-14 | Rutgers, The State University Of New Jersey | Geodate delivery vehicles |
| US9717703B2 (en) | 2009-10-16 | 2017-08-01 | Glaxosmithkline Llc | Emulsion and emulsion preconcentrate compositions comprising omega-3 fatty acids and uses thereof are disclosed |
| RU2639482C2 (en) * | 2010-12-13 | 2017-12-21 | Новартис Аг | Pharmaceutical compositions |
| US10070643B2 (en) | 2013-03-28 | 2018-09-11 | The Trustees Of Columbia University In The City Of New York | Reperfusion with omega-3 glycerides promotes donor organ protection for transplantation |
| US10842770B2 (en) | 2010-05-03 | 2020-11-24 | Teikoku Pharma Usa, Inc. | Non-aqueous taxane pro-emulsion formulations and methods of making and using the same |
| US11260018B2 (en) | 2015-09-17 | 2022-03-01 | Jrx Biotechnology, Inc. | Approaches for improving skin hydration and moisturization |
| US20220202768A1 (en) * | 2018-12-28 | 2022-06-30 | Cheers Health, Inc. | Dihydromyricetin nanoemulsion formulations and methods for forming them |
| WO2022156836A1 (en) * | 2021-01-22 | 2022-07-28 | Oncora S.R.O. | Microemulsion preconcentrate containing cladribine particularly for oral administration and method of preparation |
| US11400048B2 (en) | 2014-06-25 | 2022-08-02 | Synergia Bio Sciences Private Limited | Pharmaceutical oil-in-water nano-emulsion |
| US11465107B2 (en) * | 2019-01-31 | 2022-10-11 | Hong Ngoc Thi Dang | Process for producing a nano omega-3 microemulsion system |
| US20230158018A1 (en) * | 2018-09-13 | 2023-05-25 | Ftf Pharma Private Limited | Non-aqueous chemotherapeutic suspensions for oral dosage |
Families Citing this family (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| PT1748756E (en) * | 2004-03-10 | 2009-07-20 | Bayer Schering Pharma Ag | Compositions comprising drospirenone molecularly dispersed |
| WO2005089714A1 (en) * | 2004-03-24 | 2005-09-29 | Takeda Pharmaceutical Company Limited | Emulsion stabilizer |
| SG155189A1 (en) * | 2004-08-06 | 2009-09-30 | Transform Pharmaceuticals Inc | Novel fenofibrate formulations and related methods of treatment |
| US20070036831A1 (en) * | 2005-08-09 | 2007-02-15 | Nanobio Corporation | Nanoemulsion compositions having anti-inflammatory activity |
| WO2007041440A2 (en) | 2005-10-03 | 2007-04-12 | The Brigham And Women's Hospital, Inc. | Anti-inflammatory actions of neuroprotectin d1/protectin d1 and its natural stereoisomers |
| CN101547687A (en) * | 2005-11-17 | 2009-09-30 | 生物药效率有限公司 | Biocompatible latent emulsifiers |
| AU2008290536B2 (en) * | 2007-08-21 | 2012-02-09 | Basilea Pharmaceutica Ag | Antifungal composition |
| US20090130198A1 (en) * | 2007-11-21 | 2009-05-21 | Innopharmax Inc. | Pharmaceutical composition with enhanced bioavailability |
| JP2011006380A (en) * | 2009-05-22 | 2011-01-13 | Mochida Pharmaceut Co Ltd | EMULSIFIED COMPOSITION OF ω3 FATTY ACID |
Citations (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2372635A1 (en) * | 1976-12-03 | 1978-06-30 | Scherer Ltd R P | Pharmaceutical compsn. contg. self-emulsifying oil e.g. silicone - for admin. as capsules, giving rapid gastrointestinal resorption |
| WO1994005298A1 (en) * | 1992-08-28 | 1994-03-17 | Pharmos Corporation | Submicron emulsions as ocular drug delivery vehicles |
| WO1995008983A1 (en) * | 1993-09-30 | 1995-04-06 | Gattefosse S.A. | Oral delivery composition forming a microemulsion |
| EP0650721A1 (en) * | 1993-07-01 | 1995-05-03 | Hanmi Pharm. Ind. Co., Ltd. | Cyclosporin soft capsule |
| WO1995012384A1 (en) * | 1993-11-02 | 1995-05-11 | Yissum Research Development Company Of The Hebrew University Of Jerusalem | Self-emulsible formulation forming an oil-in-water emulsion |
| WO1995025504A1 (en) * | 1994-03-18 | 1995-09-28 | Pharmavene, Inc. | Emulsified drug delivery systems |
| WO1996013273A1 (en) * | 1994-10-26 | 1996-05-09 | Novartis Ag | Pharmaceutical compositions |
| EP0760237A1 (en) * | 1995-08-30 | 1997-03-05 | Cipla Limited | Oil-in-water microemulsions |
| US5639724A (en) * | 1984-07-24 | 1997-06-17 | Sandoz Ltd. | Cyclosporin galenic forms |
| WO1997036610A1 (en) * | 1996-07-26 | 1997-10-09 | Chong Kun Dang Corp. | Cyclosporin-containing pharmaceutical composition |
| FR2753376A1 (en) * | 1996-09-18 | 1998-03-20 | Synthelabo | PHARMACEUTICAL COMPOSITIONS COMPRISING AMISULPRIDE AND THERAPEUTIC APPLICATIONS THEREOF |
| WO1999029300A1 (en) * | 1997-12-10 | 1999-06-17 | Rtp Pharma Inc. | Self-emulsifying fenofibrate formulations |
| WO1999029316A1 (en) * | 1997-12-10 | 1999-06-17 | Severson, Mary, L. | Pharmaceutical compositions containing an omega-3 fatty acid oil |
-
1999
- 1999-05-07 JP JP2000546754A patent/JP2002513750A/en active Pending
- 1999-05-07 WO PCT/IE1999/000031 patent/WO1999056727A2/en not_active Application Discontinuation
- 1999-05-07 AU AU38439/99A patent/AU3843999A/en not_active Abandoned
- 1999-05-07 EP EP99921098A patent/EP1075252A2/en not_active Withdrawn
- 1999-05-07 CA CA002331640A patent/CA2331640A1/en not_active Abandoned
Patent Citations (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2372635A1 (en) * | 1976-12-03 | 1978-06-30 | Scherer Ltd R P | Pharmaceutical compsn. contg. self-emulsifying oil e.g. silicone - for admin. as capsules, giving rapid gastrointestinal resorption |
| US5639724A (en) * | 1984-07-24 | 1997-06-17 | Sandoz Ltd. | Cyclosporin galenic forms |
| WO1994005298A1 (en) * | 1992-08-28 | 1994-03-17 | Pharmos Corporation | Submicron emulsions as ocular drug delivery vehicles |
| EP0650721A1 (en) * | 1993-07-01 | 1995-05-03 | Hanmi Pharm. Ind. Co., Ltd. | Cyclosporin soft capsule |
| WO1995008983A1 (en) * | 1993-09-30 | 1995-04-06 | Gattefosse S.A. | Oral delivery composition forming a microemulsion |
| WO1995012384A1 (en) * | 1993-11-02 | 1995-05-11 | Yissum Research Development Company Of The Hebrew University Of Jerusalem | Self-emulsible formulation forming an oil-in-water emulsion |
| WO1995025504A1 (en) * | 1994-03-18 | 1995-09-28 | Pharmavene, Inc. | Emulsified drug delivery systems |
| WO1996013273A1 (en) * | 1994-10-26 | 1996-05-09 | Novartis Ag | Pharmaceutical compositions |
| EP0760237A1 (en) * | 1995-08-30 | 1997-03-05 | Cipla Limited | Oil-in-water microemulsions |
| WO1997036610A1 (en) * | 1996-07-26 | 1997-10-09 | Chong Kun Dang Corp. | Cyclosporin-containing pharmaceutical composition |
| FR2753376A1 (en) * | 1996-09-18 | 1998-03-20 | Synthelabo | PHARMACEUTICAL COMPOSITIONS COMPRISING AMISULPRIDE AND THERAPEUTIC APPLICATIONS THEREOF |
| WO1999029300A1 (en) * | 1997-12-10 | 1999-06-17 | Rtp Pharma Inc. | Self-emulsifying fenofibrate formulations |
| WO1999029316A1 (en) * | 1997-12-10 | 1999-06-17 | Severson, Mary, L. | Pharmaceutical compositions containing an omega-3 fatty acid oil |
Non-Patent Citations (2)
| Title |
|---|
| CRAIG D Q M ET AL: "A COMPARISON OF THE SELF-EMULSIFYING PROPERTIES OF POLYGLYCOLYSED GLYCERIDE OILS" PHARMACEUTICAL SCIENCES,GB,LONDON, vol. 1, no. 12, page 559-561 XP002053051 ISSN: 1356-6881 * |
| SHAH N H ET AL: "SELF-EMULSIFYING DRUG DELIVERY SYSTEMS (SEDDS) WITH POLYGLYCOLYZED GLYCERIDES FOR IMPROVING IN VITRO DISSOLUTION AND ORAL ABSORPTION OF LIPOPHILIC DRUGS" INTERNATIONAL JOURNAL OF PHARMACEUTICS,NL,AMSTERDAM, vol. 106, no. 1, page 15-23 XP000561369 ISSN: 0378-5173 * |
Cited By (108)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1158959A1 (en) | 1999-02-26 | 2001-12-05 | Lipocine, Inc. | Compositions and methods for improved delivery of hydrophobic therapeutic agents |
| WO2001028520A1 (en) * | 1999-10-20 | 2001-04-26 | Vesifact Ag | Microemulsion preconcentrates, microemulsion and use of such a composition |
| WO2001028518A1 (en) * | 1999-10-20 | 2001-04-26 | Vesifact Ag | Microemulsion preconcentrates which contain cyclosporines and corresponding microemulsions |
| WO2001028519A1 (en) * | 1999-10-20 | 2001-04-26 | Vesifact Ag | Microemulsion preconcentrates and microemulsions |
| US8158134B1 (en) | 1999-10-20 | 2012-04-17 | Vesifact Ag | Microemulsion preconcentrate, microemulsion and use thereof |
| EP1762557A2 (en) | 2000-02-16 | 2007-03-14 | The Brigham And Women's Hospital, Inc. | Aspirin-triggered lipid mediators |
| EP1762557A3 (en) * | 2000-02-16 | 2007-11-07 | The Brigham And Women's Hospital, Inc. | Aspirin-triggered lipid mediators |
| US7709669B2 (en) | 2000-02-16 | 2010-05-04 | The Brigham And Women's Hospital, Inc. | Aspirin-triggered lipid mediators |
| WO2001066088A1 (en) * | 2000-03-08 | 2001-09-13 | Astrazeneca Ab | New self emulsifying drug delivery system |
| US7736666B2 (en) | 2000-03-08 | 2010-06-15 | Nicox S.A. | Self emulsifying drug delivery system |
| CZ303524B6 (en) * | 2000-03-08 | 2012-11-14 | Nicox S.A. | Self-emulsifying system for medicament administration |
| AU2001237876B2 (en) * | 2000-03-08 | 2005-02-17 | Nicox S.A | New self emulsifying drug delivery system |
| JP2003525893A (en) * | 2000-03-08 | 2003-09-02 | アストラゼネカ・アクチエボラーグ | Novel self-emulsifying drug delivery system |
| JP2003525894A (en) * | 2000-03-08 | 2003-09-02 | アストラゼネカ・アクチエボラーグ | Novel self-emulsifying drug delivery system |
| KR100771318B1 (en) * | 2000-03-08 | 2007-10-29 | 니콕스 에스. 에이. | New self emulsifying drug delivery system |
| KR100771317B1 (en) * | 2000-03-08 | 2007-10-29 | 니콕스 에스. 에이. | New self emulsifying drug delivery system |
| WO2001066087A1 (en) * | 2000-03-08 | 2001-09-13 | Astrazeneca Ab | New self emulsifying drug delivery system |
| AU2001237875B2 (en) * | 2000-03-08 | 2005-05-12 | Nicox Science Ireland | New self emulsifying drug delivery system |
| AU771439B2 (en) * | 2000-06-08 | 2004-03-25 | Berhad, Hovid | A novel drug delivery system : formulation for fat-soluble drugs |
| EP1170003A1 (en) * | 2000-07-07 | 2002-01-09 | Hovid SDN BHD | Self-emulsifying drug delivery system for fat-soluble drugs |
| JP2004517052A (en) * | 2000-10-11 | 2004-06-10 | セフアロン・インコーポレーテツド | Modafinil compound-containing composition |
| WO2002053131A1 (en) * | 2000-12-28 | 2002-07-11 | Cll Pharma | Micellar colloidal pharmaceutical compositions containing a lipophilic active principle |
| FR2818905A1 (en) * | 2000-12-28 | 2002-07-05 | Cll Pharma | MICELLAR COLLOIDAL PHARMACEUTICAL COMPOSITIONS COMPRISING A LIPOPHILIC ACTIVE INGREDIENT |
| JP2006241167A (en) * | 2001-02-11 | 2006-09-14 | Aquanova German Solubilisate Technologies (Agt) Gmbh | Method for producing active ingredient concentrate and active ingredient concentrate |
| EP2298284A3 (en) * | 2001-02-14 | 2013-12-18 | GW Pharma Limited | Mucoadhesive pharmaceutical formulations |
| EP1249231A1 (en) * | 2001-04-12 | 2002-10-16 | Vesifact Ag | Pharmaceutical formulations comprising anti-inflammatory compounds and use thereof |
| WO2002083099A1 (en) * | 2001-04-12 | 2002-10-24 | Vesifact Ag | Pharmaceutical formulations containing anti-inflammatory active ingredients and the use of said formulations |
| US7815933B2 (en) | 2001-09-07 | 2010-10-19 | Nicox S.A. | Self emulsifying drug delivery system |
| JP2005504788A (en) * | 2001-09-07 | 2005-02-17 | アストラゼネカ・アクチエボラーグ | Novel self-emulsifying drug delivery system |
| US7741368B2 (en) | 2001-12-18 | 2010-06-22 | The Brigham And Women's Hospital, Inc. | Approach to antimicrobial host defense with molecular shields with EPA and DHA analogs |
| CN1332648C (en) * | 2001-12-27 | 2007-08-22 | Cj株式会社 | Microemulsion preconcentrate |
| WO2003055466A1 (en) * | 2001-12-27 | 2003-07-10 | Cj Corporation | Microemulsion preconcentrate |
| US7582785B2 (en) | 2002-04-01 | 2009-09-01 | University Of Southern California | Trihydroxy polyunsaturated eicosanoid derivatives |
| US8299116B2 (en) | 2002-07-30 | 2012-10-30 | Wyeth Llc | CCI-779 concentrate formulations |
| US7759395B2 (en) | 2002-08-12 | 2010-07-20 | The Brigham And Women's Hospital, Inc. | Use of docosatrienes, resolvins and their stable analogs in the treatment of airway diseases and asthma |
| US7585856B2 (en) | 2002-08-12 | 2009-09-08 | The Brigham And Women's Hospital, Inc. | Resolvins: Biotemplates for novel therapeutic interventions |
| US7872152B2 (en) | 2002-08-12 | 2011-01-18 | The Brigham And Women's Hospital, Inc. | Use of docosatrienes, resolvins and their stable analogs in the treatment of airway diseases and asthma |
| US9566237B2 (en) | 2002-11-01 | 2017-02-14 | Rutgers, The State University Of New Jersey | Geodate delivery vehicles |
| US7842791B2 (en) | 2002-12-19 | 2010-11-30 | Nancy Jean Britten | Dispersible pharmaceutical compositions |
| WO2004082625A2 (en) * | 2003-03-18 | 2004-09-30 | Advanced Medical Optics, Inc. | Self-emulsifying compositions, methods of use and preparation |
| WO2004082625A3 (en) * | 2003-03-18 | 2004-11-18 | Advanced Medical Optics Inc | Self-emulsifying compositions, methods of use and preparation |
| AU2004222295B2 (en) * | 2003-03-18 | 2008-07-17 | Advanced Medical Optics, Inc. | Self-emulsifying compositions, methods of use and preparation |
| US7879345B2 (en) | 2003-04-11 | 2011-02-01 | L'oreal | Composition containing an amphiphilic polymer, uses thereof |
| EP1466587A1 (en) * | 2003-04-11 | 2004-10-13 | L'oreal | Composition in the form of an oil-in-water emulsion and its cosmetic uses |
| FR2853527A1 (en) * | 2003-04-11 | 2004-10-15 | Oreal | COMPOSITION IN THE FORM OF OIL-IN-WATER EMULSION CONTAINING AN AMPHIPHILIC POLYMER, AND ITS PARTICULARLY COSMETIC USES |
| WO2005102277A1 (en) * | 2004-04-19 | 2005-11-03 | Hewlett-Packard Development Company, L.P. | A composition of a jetable microemulsion, a method of manufacturing thereof as well as a system of dispensing |
| US8728518B2 (en) | 2004-08-27 | 2014-05-20 | Cspc Zhongqi Pharmaceutical Technology (Shijiazhuang) Co., Ltd | Butylphthalide self-emulsifying drug delivery system, its preparation and method and application |
| US8518961B2 (en) | 2004-11-19 | 2013-08-27 | Sigma-Tau Industrie Farmaceutiche Riunite S.P.A. | Pharmaceutical compositions comprising a camptothecin derivate |
| US20140287004A1 (en) * | 2005-11-14 | 2014-09-25 | The Trustees Of Columbia University In The City Of New York | Use of an Omega-3 Lipid-Based Emulsion Following Ischemic Injury to Provide Protection and Recovery in Human Organs |
| US20150272921A1 (en) * | 2005-11-14 | 2015-10-01 | The Trustees Of Columbia University In The City Of New York | Use of an Omega-3 Lipid-Based Emulsion Following Ischemic Injury to Provide Protection and Recovery in Human Organs |
| WO2008002121A2 (en) * | 2006-06-28 | 2008-01-03 | Hovid, Berhad | A self-emulsifying composition for poorly bioavailable drugs |
| WO2008002121A3 (en) * | 2006-06-28 | 2008-09-18 | Hovid Berhad | A self-emulsifying composition for poorly bioavailable drugs |
| TWI405589B (en) * | 2006-06-28 | 2013-08-21 | Hovid Berhad | An effective pharmaceutical carrier for poorly bioavailable drugs |
| RU2480208C2 (en) * | 2006-10-04 | 2013-04-27 | М Унд П Патент Акциенгезелльшафт | Delivery system with controlled release for nasal application of neurotransmitters |
| US9339460B2 (en) | 2007-12-26 | 2016-05-17 | Virbac Sa | Self-emulsifiable liquid florfenicol composition intended to be incorporated into the drinking water of livestock |
| FR2925855A1 (en) * | 2007-12-26 | 2009-07-03 | Virbac Sa Sa | FLORFENICOL SELF-CONTAINING LIQUID COMPOSITION FOR INCORPORATING BEVERAGE WATER IN ANIMAL BREEDING ANIMALS. |
| WO2009106714A2 (en) | 2007-12-26 | 2009-09-03 | Virbac Sa | Self-emulsifiable liquid florfenicol composition intended to be incorporated into the drinking water of livestock animals |
| WO2009106714A3 (en) * | 2007-12-26 | 2009-12-17 | Virbac Sa | Self-emulsifiable liquid florfenicol composition intended to be incorporated into the drinking water of livestock animals |
| US9532963B2 (en) | 2009-03-09 | 2017-01-03 | Pronova Biopharma Norge As | Compositions comprising a fatty acid oil mixture and a free fatty acid, and methods and uses thereof |
| US10596142B2 (en) | 2009-03-09 | 2020-03-24 | Pronova Biopharm Norge AS | Compositions comprising a fatty acid oil mixture and a free fatty acid, and methods and uses thereof |
| EP2405895A4 (en) * | 2009-03-09 | 2012-10-10 | Pronova Biopharma Norge As | Compositions comprising a fatty acid oil mixture comprising epa and dha in free acid form and a surfactant, and methods and uses thereof |
| EP2405903A4 (en) * | 2009-03-09 | 2012-10-10 | Pronova Biopharma Norge As | Compositions comprising a fatty acid oil mixture and a free fatty acid, and methods and uses thereof |
| WO2010103402A1 (en) * | 2009-03-09 | 2010-09-16 | Pronova Biopharma Norge As | Compositions comprising a fatty acid oil mixture comprising epa and dha in free acid form and a surfactant, and methods and uses thereof |
| US11612579B2 (en) | 2009-03-09 | 2023-03-28 | Pronova Biopharma Norge As | Compositions comprising a fatty acid oil mixture and a surfactant, and methods and uses thereof |
| US11395811B2 (en) | 2009-03-09 | 2022-07-26 | Pronova Biopharma Norge As | Compositions comprising a fatty acid oil mixture comprising EPA and DHA in free acid form and a surfactant, and methods and uses thereof |
| WO2010103404A1 (en) * | 2009-03-09 | 2010-09-16 | Pronova Biopharma Norge As | Compositions comprising a fatty acid oil mixture and a surfactant, and methods and uses thereof |
| WO2010119319A1 (en) * | 2009-03-09 | 2010-10-21 | Pronova Biopharma Norge As | Compositions comprising a fatty acid oil mixture and a free fatty acid, and methods and uses thereof |
| US10028928B2 (en) | 2009-03-09 | 2018-07-24 | Pronova Biopharma Norge As | Compositions comprising a fatty acid oil mixture and a free fatty acid, and methods and uses thereof |
| EP3915554A1 (en) * | 2009-03-09 | 2021-12-01 | Basf As | Compositions comprising a fatty acid oil mixture and a surfactant, and methods and uses thereof |
| EA022028B1 (en) * | 2009-03-09 | 2015-10-30 | Пронова Биофарма Норге Ас | Compositions comprising a fatty acid oil mixture and a surfactant, and methods and uses thereof |
| EP2405895A1 (en) * | 2009-03-09 | 2012-01-18 | Pronova BioPharma Norge AS | Compositions comprising a fatty acid oil mixture comprising epa and dha in free acid form and a surfactant, and methods and uses thereof |
| EP2405902A4 (en) * | 2009-03-09 | 2012-10-10 | Pronova Biopharma Norge As | Compositions comprising a fatty acid oil mixture and a surfactant, and methods and uses thereof |
| EP2405903A1 (en) * | 2009-03-09 | 2012-01-18 | Pronova BioPharma Norge AS | Compositions comprising a fatty acid oil mixture and a free fatty acid, and methods and uses thereof |
| EP2405902A1 (en) * | 2009-03-09 | 2012-01-18 | Pronova BioPharma Norge AS | Compositions comprising a fatty acid oil mixture and a surfactant, and methods and uses thereof |
| EP2433630A1 (en) * | 2009-05-22 | 2012-03-28 | Mochida Pharmaceutical Co., Ltd. | Self-emulsifying composition of 3 fatty acid |
| US11207286B2 (en) | 2009-05-22 | 2021-12-28 | Mochida Pharmaceutical Co., Ltd. | Self-emulsifying composition of w3 fatty acid |
| US9089483B2 (en) | 2009-05-22 | 2015-07-28 | Mochida Pharmaceutical Co., Ltd. | Self-emulsifying composition of OMEGA3 fatty acid |
| EP2433630A4 (en) * | 2009-05-22 | 2014-01-08 | Mochida Pharm Co Ltd | Self-emulsifying composition of 3 fatty acid |
| US8618168B2 (en) | 2009-05-22 | 2013-12-31 | Mochida Pharmaceutical Co., Ltd. | Self-emulsifying composition of OMEGA3 fatty acid |
| US10092536B2 (en) | 2009-05-22 | 2018-10-09 | Mochida Pharmaceutical Co., Ltd. | Self-emulsifying composition of ω3 fatty acid |
| US10493052B2 (en) | 2009-05-22 | 2019-12-03 | Mochida Pharmaceutical Co., Ltd. | Self-emulsifying composition of ω3 fatty acid |
| US9579281B2 (en) | 2009-05-22 | 2017-02-28 | Mochida Pharmaceutical Co., Ltd. | Self-emulsifying composition of ω3 fatty acid |
| US10668038B2 (en) | 2009-10-16 | 2020-06-02 | Mochida Pharmaceutical Co., Ltd. | Emulsion and emulsion preconcentrate compositions comprising omega-3 fatty acids and uses thereof are disclosed |
| US9717703B2 (en) | 2009-10-16 | 2017-08-01 | Glaxosmithkline Llc | Emulsion and emulsion preconcentrate compositions comprising omega-3 fatty acids and uses thereof are disclosed |
| EP2490678A4 (en) * | 2009-10-23 | 2013-05-01 | Pronova Biopharma Norge As | Coated capsules and tablets of a fatty acid oil mixture |
| EP2490678A1 (en) * | 2009-10-23 | 2012-08-29 | Pronova BioPharma Norge AS | Coated capsules and tablets of a fatty acid oil mixture |
| US8932610B2 (en) | 2010-03-01 | 2015-01-13 | Laboratorios Salvat, S.A. | Aqueous clear solutions of fluocinolone acetonide for treatment of otic inflammation |
| US10842770B2 (en) | 2010-05-03 | 2020-11-24 | Teikoku Pharma Usa, Inc. | Non-aqueous taxane pro-emulsion formulations and methods of making and using the same |
| US20140017308A1 (en) * | 2010-09-08 | 2014-01-16 | Pronova Biopharma Norge As | Compositions comprising a fatty acid oil mixture, a free fatty acid, and a statin |
| WO2012032414A2 (en) * | 2010-09-08 | 2012-03-15 | Pronova Biopharma Norge As | Compositions comprising a fatty acid oil mixture, a surfactant, and a statin |
| WO2012032414A3 (en) * | 2010-09-08 | 2012-05-03 | Pronova Biopharma Norge As | Compositions comprising a fatty acid oil mixture, a surfactant, and a statin |
| WO2012032415A3 (en) * | 2010-09-08 | 2012-05-03 | Pronova Biopharma Norge As | Compositions comprising a fatty acid oil mixture comprising epa and dha in free acid form, a surfactant, and a statin |
| WO2012032417A3 (en) * | 2010-09-08 | 2012-05-03 | Pronova Biopharma Norge As | Compositions comprising a fatty acid oil mixture, a free fatty acid, and a statin |
| RU2639482C2 (en) * | 2010-12-13 | 2017-12-21 | Новартис Аг | Pharmaceutical compositions |
| US10188696B2 (en) | 2010-12-13 | 2019-01-29 | Novartis Ag | Pharmaceutical compositions |
| US9763880B2 (en) | 2012-10-01 | 2017-09-19 | Teikoku Pharma Usa, Inc. | Non-aqueous taxane formulations and methods of using the same |
| US9308195B2 (en) | 2012-10-01 | 2016-04-12 | Teikoku Pharma Usa, Inc. | Non-aqueous taxane formulations and methods of using the same |
| US8940786B2 (en) | 2012-10-01 | 2015-01-27 | Teikoku Pharma Usa, Inc. | Non-aqueous taxane nanodispersion formulations and methods of using the same |
| US11076593B2 (en) | 2013-03-28 | 2021-08-03 | The Trustees Of Columbia University In The City Of New York | Reperfusion with omega-3 glycerides promotes donor organ protection for transplantation |
| US10070643B2 (en) | 2013-03-28 | 2018-09-11 | The Trustees Of Columbia University In The City Of New York | Reperfusion with omega-3 glycerides promotes donor organ protection for transplantation |
| US10736842B2 (en) | 2014-06-25 | 2020-08-11 | Synergia Bio Sciences Private Limited | Pharmaceutical oil-in-water nano-emulsion |
| US11400048B2 (en) | 2014-06-25 | 2022-08-02 | Synergia Bio Sciences Private Limited | Pharmaceutical oil-in-water nano-emulsion |
| WO2015198350A1 (en) * | 2014-06-25 | 2015-12-30 | Synergia Bio Sciences Private Limited | A pharmaceutical oil-in-water nano-emulsion |
| US11260018B2 (en) | 2015-09-17 | 2022-03-01 | Jrx Biotechnology, Inc. | Approaches for improving skin hydration and moisturization |
| US20230158018A1 (en) * | 2018-09-13 | 2023-05-25 | Ftf Pharma Private Limited | Non-aqueous chemotherapeutic suspensions for oral dosage |
| US20220202768A1 (en) * | 2018-12-28 | 2022-06-30 | Cheers Health, Inc. | Dihydromyricetin nanoemulsion formulations and methods for forming them |
| US11465107B2 (en) * | 2019-01-31 | 2022-10-11 | Hong Ngoc Thi Dang | Process for producing a nano omega-3 microemulsion system |
| WO2022156836A1 (en) * | 2021-01-22 | 2022-07-28 | Oncora S.R.O. | Microemulsion preconcentrate containing cladribine particularly for oral administration and method of preparation |
Also Published As
| Publication number | Publication date |
|---|---|
| CA2331640A1 (en) | 1999-11-11 |
| AU3843999A (en) | 1999-11-23 |
| EP1075252A2 (en) | 2001-02-14 |
| WO1999056727A3 (en) | 2000-03-02 |
| JP2002513750A (en) | 2002-05-14 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AU743098B2 (en) | Pharmaceutical compositions containing an omega-3 fatty acid oil | |
| EP1075252A2 (en) | Solvent/cosolvent free microemulsion and emulsion preconcentrate drug delivery systems | |
| EP1035862B1 (en) | Cyclosporin-containing microemulsion preconcentrate composition | |
| JP3391961B2 (en) | Cyclosporin-containing soft capsule composition | |
| SK52197A3 (en) | Pharmaceutical compositions | |
| JP2002505271A (en) | Emulsion pre-concentrate containing cyclosporins or macrolides | |
| CA2283780A1 (en) | Lipophilic binary systems for the administration of lipophilic compounds | |
| US6028067A (en) | Cyclosporin-containing microemulsion preconcentrate composition | |
| EP1413297A1 (en) | Cyclosporin-containing soft capsule preparations | |
| KR100300251B1 (en) | Cyclosporine-containing pharmaceutical composition | |
| AU741923B2 (en) | Pharmaceutical compositions | |
| KR20070096142A (en) | Nanoemulsion containing tacrolimus for increasing the absorption and the preparation thereof | |
| MXPA00005507A (en) | Cyclosporin-containing microemulsion preconcentrate composition |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AL AM AT AU AZ BA BB BG BR BY CA CH CN CU CZ DE DK EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MD MG MK MN MW MX NO NZ PL PT RO RU SD SE SG SI SK SL TJ TM TR TT UA UG US UZ VN YU ZA ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): GH GM KE LS MW SD SL SZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| AK | Designated states |
Kind code of ref document: A3 Designated state(s): AE AL AM AT AU AZ BA BB BG BR BY CA CH CN CU CZ DE DK EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MD MG MK MN MW MX NO NZ PL PT RO RU SD SE SG SI SK SL TJ TM TR TT UA UG US UZ VN YU ZA ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A3 Designated state(s): GH GM KE LS MW SD SL SZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1999921098 Country of ref document: EP |
|
| ENP | Entry into the national phase in: |
Ref document number: 2331640 Country of ref document: CA |
|
| NENP | Non-entry into the national phase in: |
Ref country code: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 38439/99 Country of ref document: AU |
|
| WWP | Wipo information: published in national office |
Ref document number: 1999921098 Country of ref document: EP |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 1999921098 Country of ref document: EP |